CRYOTECHNICS
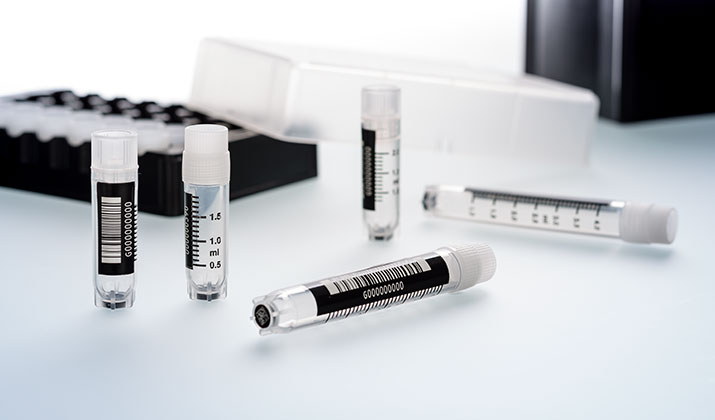
For sample freezing and storage, Greiner Bio-One offers a comprehensive product portfolio including Cryo.s tubes, racks as well as electronic equipment for tube closure and sample tracking. With the brand Cryo.s Greiner Bio-One combines more than 30 years of experience in the manufacturing of cryogenic tubes with latest technology and innovation.
Cryo.s, Cryo.s with Datamatrix and linear barcode and Cryo.s Biobanking tubes are for storage of tissue, cells, fungi, bacteria, spores, cellular extracts or body fluids at ultra-low temperature for research and development purposes as well as in-vitro diagnostics.
Sub Categories
Sign up to our newsletter or request more information here